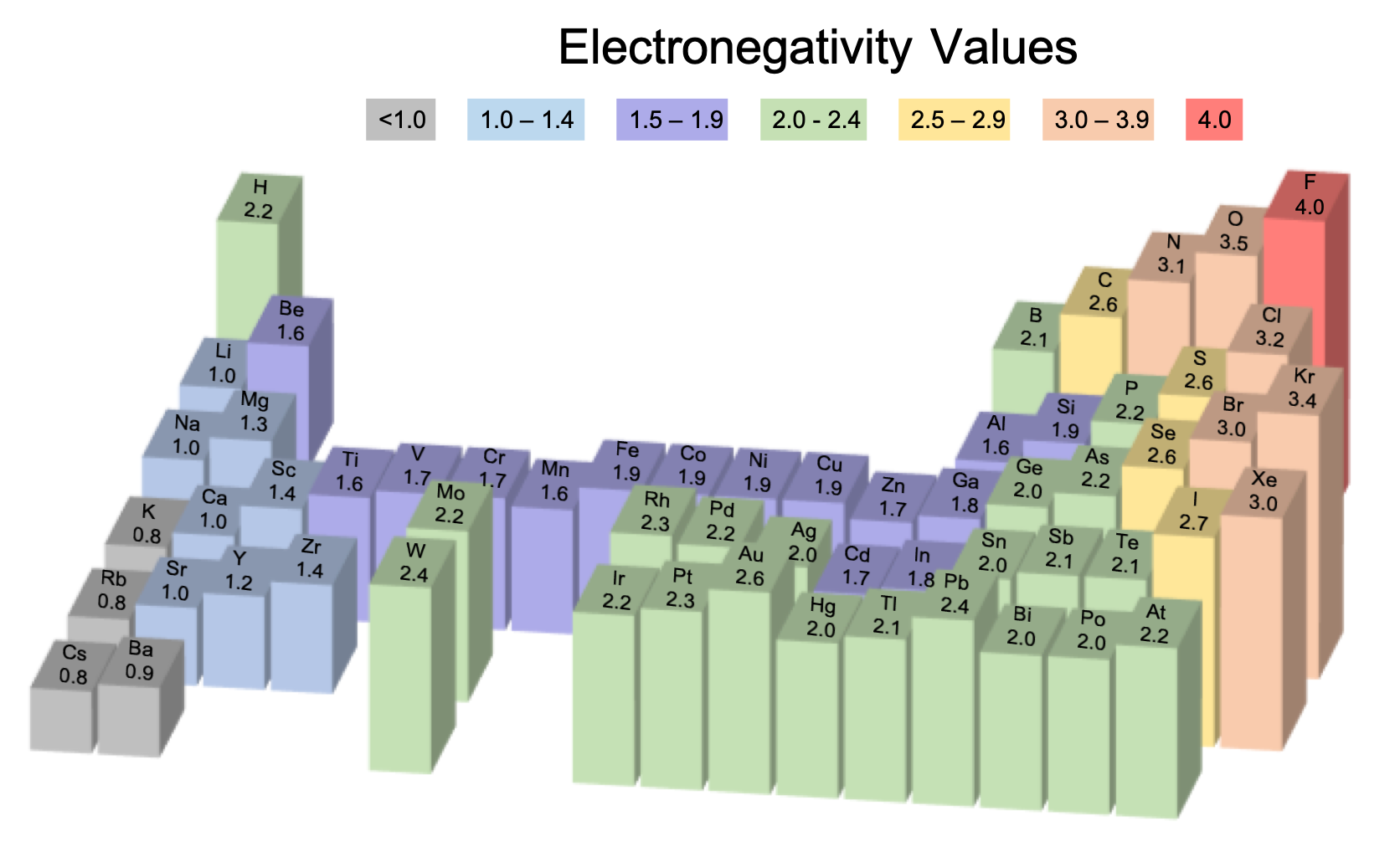
Electronegativity increases as we move from left to right across a period. From this comparison, we can describe the horizontal trend for electronegativity on the periodic table. So it is much more likely to gain electrons or attract them. 
On the other hand, bromine is just one electron away from filling that outer electron shell. Potassium with its one valence electron is more likely to give up that electron and is unlikely to gain or attract electrons. Meanwhile, bromine with seven valence electrons has a nearly full outer electron shell. Potassium with just one valence electron has a nearly empty outermost electron shell. One significant difference between potassium and bromine is the number of valence electrons. Let’s start analyzing the horizontal trend by looking at two elements that are on opposite ends of the same period. In other words, how does electronegativity change as we move side to side or up and down on the periodic table? If we can understand these two directional trends, we can find the corner of the periodic table that is likely to have the highest electronegativity values. This question is asking us to look at the periodic table and find the trends in electronegativity. As a result, those electrons pull in closer to the oxygen atom than to the hydrogen atoms. Since the oxygen atom is more electronegative than the hydrogen atoms, it has a stronger attraction to the bonding pairs of electrons. For example, the covalent bonds that hold together a water molecule represent a pair of shared electrons. Electronegativity is defined as the tendency of an atom to attract a bonding pair of electrons. This question is about electronegativity. 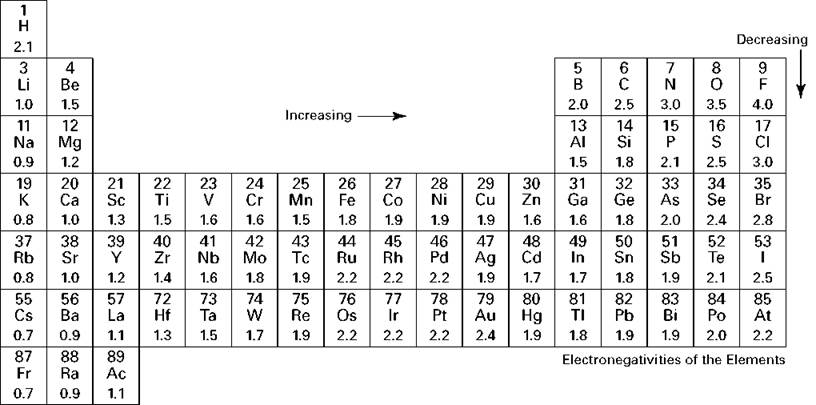
Which letter corresponds to the elements in the periodic table that are the most electronegative?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
